Technical lecture by gempex
ECA Live Online Training on 13. December 2022
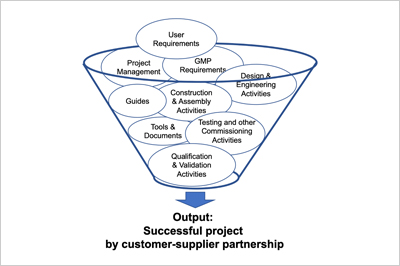
In this training, the "ECA Good Practice Guide Integrated Qualification and Validation" is presented for practical use. The required close cooperation between customers and suppliers is in the foreground with the aim to consider qualification and validation together and to lead them to success. Good Engineering Practice (GEP) is integrated into the qualification activities. One of the essential tools is risk analysis, which is now also introduced in qualification from the very beginning - from the creation of the project URS right through to performance qualification, the PQ. Q&V Case Studies round off the program, they impressively show how worthwhile it is to work together with suppliers.
The ECA Good Practice Guide was developed over a period of 4 years in close cooperation between companies in the pharmaceutical industry, plant manufacturers and their suppliers for process technology. Version 2.2, which is now available, was recently published. It provides a good basis for action for anyone whose projects are affected by Annex 15 of the EU GMP Guideline or the Process Validation Guideline of the FDA.
Overview: Integrated Qualification and Validation
Ralf Gengenbach, Managing Director gempex GmbH, as moderator of the event, first presents the Good Practice Guide in its entirety, explains the history and the formerly recognized need to get one more guideline. A comparison with other sets of rules is given. With the ECA Good Practice Guide, the focus is newly placed on a successful customer-supplier relationship.
The further program will present practical examples that demonstrate the central role played by lifecycle documents that are cleanly prepared from the outset. In addition, real challenges that can arise from individual project contracts and their implementation will be discussed.
More detailed information and the possibility of registration are provided at the organizer, the ECA Academy.
The gempex expert team will be happy to answer any questions on this topic via contact@gempex.com.